
Boson covid test
$4.18 Original price was: $4.18.$2.19Current price is: $2.19.
As Low As $1.75 per test
Authorized by FDA under an Emergency Use Authorization (EUA)
SHIPPING SAME DAY
| Test info | |
| Covid test | |
| Nucleocapsid Protein Antigen | |
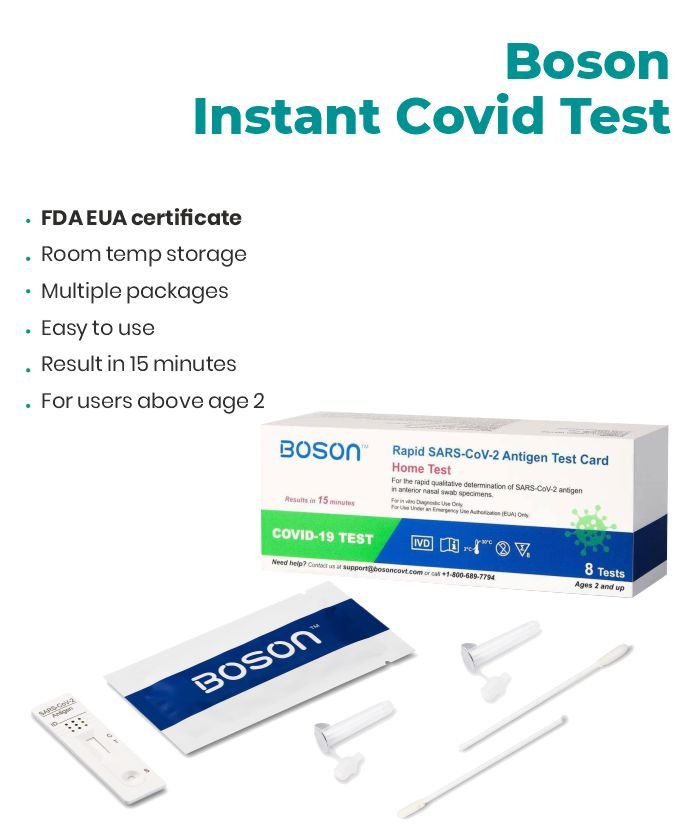
Rapid Results at Your Fingertips: Introducing the Boson COVID Antigen Test, FDA COVID test for At-Home Use
In the ever-evolving landscape of healthcare, the need for accessible and efficient COVID-19 testing remains paramount. The boson covid test emerges as a beacon of hope, providing a reliable and instant solution for individuals seeking peace of mind in the comfort of their homes. With approval from the FDA, this at home covid test is a game-changer in the fight against the pandemic.
The Power of Instant Results
The Boson covid test is designed with convenience in mind, offering users the ability to receive rapid results without the hassle of scheduling appointments or waiting in long lines at testing centers. By bringing the testing process to the home front, individuals can now take charge of their health with ease and speed.
FDA Approval: A Mark of Trust
Rest assured, the Boson COVID Antigen Test has earned the stamp of approval from the U.S. Food and Drug Administration (FDA). This stringent regulatory validation ensures the test’s accuracy, reliability, and safety, providing users with the confidence they need during these uncertain times.
The FDA’s rigorous evaluation process means you can trust the rapid covid test results delivered by the Boson test.
Why Choose Boson?
- Reliability: With FDA approval, the Boson COVID Antigen Test has undergone rigorous testing to ensure accuracy in detecting the virus, providing users with confidence in their results.
- Convenience: No need to venture outside or endure long wait times. The Boson test can be administered in the privacy of your home, saving you time and effort.
- Speed: Instant results mean quick decision-making. Whether for travel, family gatherings, or personal peace of mind, the Boson covid test delivers swift answers.
- Accessibility: The Boson test is a cost-effective and easily accessible solution for individuals looking to prioritize their health without compromising on quality.
In the ongoing battle against COVID-19, the Boson COVID Antigen Test stands as a beacon of hope, offering a reliable, at-home solution that has received the coveted FDA approval.
With its user-friendly design, swift results, and commitment to accuracy, the Boson test is a powerful tool for individuals seeking to navigate the challenges of the pandemic with confidence and ease.
Embrace the power of instant results and take control of your health with the Boson covid test.
Test Procedure Instant covid test
The test procedure overview does not replace the package insert. Please read and follow the detailed instructions in the Quick Reference Instructions.
1.Swab
Slowly rotate the swab in a circular motion 5 times by firmly pressing against the inside walls of the nostril for a total of 15 seconds. Do not just spin the swab. Gently remove the swab and repeat in the other nostril using the same swab.

2. Dip
Place the swab into the buffer tube. Rotate the swab 5 times. Set a timer and leave the swab in the buffer tube for 1 minute. Pinch the buffer tube with your fingers and remove the solution from the swab as much as possible.

3. Drop
Invert the buffer tube and add 3 drops of the test sample into the sample well (S) by gently squeezing the extraction tube. Do not add the test sample to the rectangular results window.

Reading the results of BOSON rapid sarscov antigen test

Negative result
If the Control (C) line is visible, but the Test (T)line is not visible, the test is negative.
A negative rapid covid test result indicates that antigens from the virus that causes COVID-19 were not detected from the specimen. A negative result does not rule out COVID-19. There is a higher chance of false negative results with antigen tests than with laboratory-based molecular tests. This means that there is a higher chance this test will give you a negative result when you have COVID-19.

If a Control (C) line and the Test (T) line are visible, the test is positive. Any faint visible pink/purple test (T) line with the control line (C) should be read as positive.
A positive rapid covid test result means that the virus that causes COVID-19 was detected in your sample and it is very likely you have COVID-19 and are contagious. Please contact your doctor/primary care physician or your local health authority immediately and adhere to the local guidelines regarding self-isolation.

If a control line (C) is not visible, even if the test line is visible, the result must be considered invalid.
An invalid result means the test was not able to tell if you have COVID-19 or not. If the test is invalid, a new rapid covid test kit should be used to collect a new nasal specimen and the test should be run again, using a new test and tube.
Covid Antigen Home Test
- A rapid test for the detection of SARS-CoV-2 antigens in anterior nasal specimens.
- For Emergency Use Authorization (EUA) use only.
- In vitro diagnostic use only.
- For more information on EUAs visit: https://www.fda.gov/emergency-preparedness-and-response/mcm-legal-regulatoryand-policy-framework/emergency-use-authorization
- For the most up to date information on COVID-19, please visit: www.cdc.gov/COVID19
COVID-19 is an acute respiratory infectious disease caused by the SARS-CoV-2 virus, a novel Betacoronavirus. SARS-CoV-2 is mostly spread person-to-person, both by individuals with symptoms of COVID-19 infection and by infected people without symptoms.
Based on the current knowledge, the incubation period is 1 to 14 days, mostly 4-5 days. Symptoms include fever, fatigue, and cough. For a full list of symptoms, see: https://www.cdc.gov/coronavirus/2019- ncov/symptoms-testing/symptoms.html.
If no control line shows up on the test, the result is invalid (even if any test line shows up). An invalid result means the test was not able to tell if you have COVID-19 or not. If the test is invalid, a new swab should be used to collect a new nasal specimen and the test should be run again, using all new test components.
No, the nasal swab is not sharp, and it should not hurt. Sometimes the swab can feel slightly uncomfortable. If you feel pain, please stop the test and seek advice from your healthcare provider.
There are different kinds of tests for COVID-19. Molecular tests (also known as PCR tests) detect genetic material from the virus. Antigen tests, such as the Rapid SARS-CoV-2 Antigen Test detect proteins from the virus.
Antigen tests are very specific for the COVID-19 virus but are not as sensitive as molecular tests.
This means that a positive result is highly accurate, but a negative result does not rule out infection. If your test result is negative, you should discuss with your healthcare provider whether an additional molecular test is necessary and if you should continue isolating at home.
The performance of the Rapid SARS-CoV-2 Antigen Test Card was established in a prospective clinical study of symptomatic individuals using an EUA molecular test as a comparator method.
The data from this study were analyzed using the minimum recommended number of low positives demonstrating that the test correctly identified 82.7% of positive samples and correctly identified 99.1% of negative samples.
For more detailed information on test performance please see Section 2.6 of the Health Care Provider Instructions for Use.
| Weight | 0.8 lbs |
|---|---|
| Dimensions | 6 × 5 × 3.5 in |
You must be logged in to post a review.








Reviews
There are no reviews yet.